ABOUT DESITIN
- Home
- /
- Promoted products
Promoted Products
Desitin is focused on pharmaceuticals for the treatment of epilepsy. It is the only manufacturer that offers the almost entire spectrum of products recommended by the WHO for the treatment of epilepsy. It should be noted that not all products are available in all countries.
Desitin not only offers a wide range of different therapeutics, but also a broad spectrum of presentations and dosage strengths.
Whereas most of these preparations have been developed in-house by Desitin and successfully sold within various countries of Europe, co-operations with licensors and the successful marketing of products licensed represent a cornerstone of Desitin’s latest developments.
Among the products which are presently in the centre of our marketing efforts four products, namely Xeomin , Timox, Clarium and Diacomit, are licensed from European partner companies (Merz, Servier, Biocodex). These products are not currently available in the UK.
Besides its full range of antiepileptics Desitin promotes the following proprietary and licensed products:
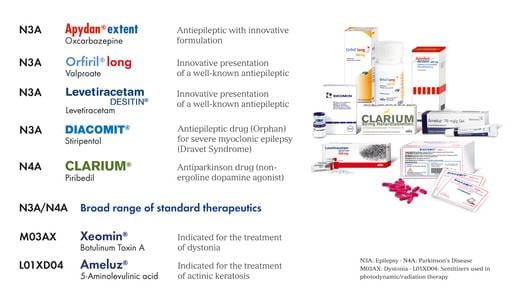
Note: In the UK Orfiril long and Orfiril solution for injection are marketed under the brand name of Episenta®▼ (sodium valproate prolonged release) and Episenta®▼ Solution for Injection.
In the UK Levetiracetam Desitin is marketed under the brand name of Desitrend® (levetiracetam)
None of the other products shown are available in the UK at this time from Desitin.